This is one of my
space related projects. The status of this
document is
work in progress.
Are Amateur Orbital Rockets Possible?
Please note that this document was originally written
in 1995 with only minor updates moving forward. As
such, it is getting pretty dated.
Table of Contents
Even the best designed rockets sometimes explode,
and when they do, the high speed metal fragments
that are ejected from the explosion can be thought
of as shrapnel that can cause grievous harm if any
pieces hit anyone. Since this a paper about about
big amateur rockets, explosions and the
resulting shrapnel are extremely likely. If you
decide to try out anything suggested in this document,
you do so at your own risk, and with absolutely no
liability to me. If this is unacceptable to you,
do not attempt anything described herein. Since
I value my life, you can rest assured that I will
not be anywhere near any of the equipment described
in this document when it is being tested.
This document is basically a bunch of notes to
myself about the issues involved in designing
a fairly big amateur rocket that is capable
inserting a small payload into low earth orbit.
Think of this document as a notes in a design
journal and you have the right basic idea.
You should definitely not read this document as
a paper written by anybody authoritative -- I'm
strictly an armchair `wanna be' rocket scientist
at the moment. My training is in electrical
engineering and computer science, not aerospace
engineering. Some of the content in this
document is undoubtedly wrong
and I would appreciate if people more knowledgeable
than me who read it could
send me some E-mail pointing out any mistakes.
I am very impressed with what the amateur rocket
community has been able to build and launch to date.
The recent launch of a 14000 pound thrust rocket to
an altitude of approximately 50 miles by the Reaction
Rocket Society and written up by David Crisalli
[Crisalli97] is one such recent example.
As impressive as the amateur rocket community's
accomplishments have been, building a rocket that
is capable of launching even the smallest of payloads
into orbit has been beyond the financial and
technical means of the amateur rocket community.
This paper explores some ideas on how to expand
the envelope of amateur rockets to include the
insertion of small payloads into orbit.
The amateur rocket community has been attracted
to solid propellant rockets due to their innate
simplicity and relative low cost. While a
solid rocket is technically capable of putting
a payload into orbit, it has to be pretty big
multi-stage solid rocket to do so. The large size
and multiple stages introduce cost, safety, and
control issues that, to date, have not been
surmounted by the amateur rocket community.
Liquid fuel engines are quite a bit more complicated
to manufacture and test, but they have improved
performance, safety and control capabilities over
and above solid propellant rockets. Are liquid
fueled rocket engines feasible to the amateur
community? Some early experimentation with amateur
liquid-fueled rocket engines is written up by Leroy
Krzycki in a small book entitled How to Design,
Build, and Test Small Liquid-Fueled Rocket Engines
[Krzycki67]. This book describes at a reasonable
level of detail the issues involved with designing,
building, and testing small (10-20 pound thrust)
liquid-fueled amateur rocket engines. Can such
small engines be scaled up by the amateur community
to be capable of putting a small payload into
low earth orbit?
The ideal fuel/oxidizer combination for amateur
rockets would have low toxicity, easy handling
characteristics, low cost, and high performance
(high Isp.) Alas, there is no such perfect
fuel/oxidizer combination. The selection of
a fuel/oxidizer combination is an exercise in
compromises. So what characteristics are most
important to amateur community? Well, I would
propose that the characteristics should be ordered
by low fume toxicity first, reasonable
handling/cost/availability second, and reasonable
performance last. By low fume toxicity, I mean
that the fumes given off by the substance are not
too poisonous. By this criterion, gasoline is
considered reasonable and hydrazine is not. In
the discussion below, I will simply leave the high
fume toxicity fuels and oxidizers off the list;
this shortens the oxidizer and fuel lists down
considerably.
The list of reasonable fume toxicity oxidizers
is quite short:
-
Hydrogen Peroxide (H2O2)
-
Hydrogen peroxide is probably a pretty
marginal oxidizer for the amateur rocket
community. Hydrogen peroxide can be
made to react with itself
(2H2O2 =>
2H2O + 2O2 +
energy). Thus, hydrogen peroxide can be
used as a monopropellant (like hydrazine.)
I am not really all that familiar with
the steps that are needed to prevent
hydrogen peroxide from reacting with
itself. If the steps are not too onerous,
it is possible to use it as an oxidizer
in a bi-propellant system as well.
Hydrogen peroxide is not readily available
in any significant quantity or purity
for the amateur rocket community.
The hydrogen peroxide sold by the corner
drug store is heavily diluted with water.
This forces the amateur into developing
the technology to manufacture or purify
hydrogen peroxide. This can make the
cost per liter of purified hydrogen
peroxide quite expensive.
-
Nitrous Oxide (N2O)
-
Nitrous oxide is a liquid at ambient temperature
and several atmospheres of pressure. This
makes nitrous oxide a little more difficult
to handle than hydrogen peroxide, but it is
still reasonable. Like hydrogen peroxide,
nitrous oxide is a bit difficult to obtain.
It is used by the dental community for mild
anesthesia. The abuse of nitrous oxide at
parties has caused some restrictions on
obtaining bottles of gaseous nitrous oxide.
The cost per liter of purified nitrous oxide
can be quite expensive.
-
Oxygen (O2)
-
Oxygen is both inexpensive and readily
available in both gaseous and liquid form
due to its use by the welding industry.
Gaseous oxygen can be used in the early
development of an amateur rocket engine.
Use of liquid oxygen (LOX) is necessary for
a flight weight vehicle. LOX is more
difficult to handle than the other ambient
temperature oxidizers mentioned above.
The difficulty with LOX is that it can
cause spontaneous combustion when it
comes in contact with various substances --
like finger print grease! Thus, handling
LOX requires a level of cleanliness that
can be difficult to obtain in the amateur
environment.
and the list of reasonable fume toxicity fuels is a
basically a bunch of different hydrocarbons (and
plain old hydrogen):
-
Hydrogen (H2)
-
Hydrogen is not readily available, but it is
both easy and inexpensive to electrolyze from
water. Liquid hydrogen (LH2) is an extremely
cold cryogenic fluid, that requires very careful
handling. A pipe of LH2 exposed to air will
cause the air around the pipe to liquidize. Thus,
pipes carrying liquid hydrogen tend to vacuum
jacketed. Another disadvantage of LH2 is that
it is not very dense, thereby necessitating
fairly large tanks in comparison to the oxidizer
tank. LH2 has some truly awesome performance
characteristics that can offset its handling
disadvantages. Frankly, LH2 is probably too
difficult to handle for the amateur rocket
community.
-
Methane (CH4)
-
Methane is basically the primary component in
natural gas. While liquidized natural gas
(LNG) is not that readily available, it is
possible to create LNG by simply hooking a
tube up to natural gas tube and running the
the gas through a heat exchanger consisting
of a pipe immersed in liquid nitrogen. Given
that both butane and propane are readily
available and easier to liquidize, it is probably
easier to use either of those fuels instead
of LNG. LNG appears to have a higher performance
than either butane or methane.
-
Propane (C3H8)
-
Like butane, propane is readily available and
quite inexpensive due to extensive use in the
recreational vehicle industry. Propane has an
interesting characteristic in that it freezes at
a temperature that is below that the boiling point
of LOX. This can really help simplify the
tank design issues when dealing with LOX.
Butane (C4H10) -
Butane is both readily available and relatively
inexpensive due to its use for rural home heating.
Butane is a liquid under a pressure of several
atmospheres. Butane has one disadvantage in
comparison to propane in that it is a frozen
solid at the same temperature that LOX boils.
-
Gasoline (C8H18)
-
Gasoline is extremely available and fairly low
cost due to its use by cars and trucks. It is
a liquid at both ambient temperature and pressure.
The handling characteristics of gasoline are
quite well understood.
-
Kerosene (C?H?)
-
Kerosene is not quite as available as gasoline,
but it can be obtained and it basically has
similar performance characteristics as gasoline.
-
Methyl Alcohol (CH3OH)
-
Methyl alcohol is another viable alternative
to gasoline and kerosene. Methyl alcohol is
not as available as either kerosene or gasoline.
-
Ethyl Alcohol (CH3CH2OH)
-
Actually, ethyl alcohol (i.e.grain alcohol,
booze, etc.) is not very expensive when you
remove the taxes. If you are willing to go
through the hassle of convincing the appropriate
authorities that you are going to burn it
instead of drink it, you can get a waiver on
paying the taxes. Be prepared to explain why
some other fuel will not suffice though.
I probably missed a few fuels and oxidizers that could
be considered for amateur use. The more toxic stuff
(e.g. red fuming nitric acid, hydrazine, fluorine, etc.)
has definitely been left off. I also left off some
pretty reasonable fuels like JP-4 (Jet Propellant 4)
and RP-4 (Rocket Propellant 4) because they are a
little difficult to obtain. It might be possible to
obtain JP-4 from your favorite airport if you think
you can convince someone to sell it to you in the
quantities you need. Obtaining RP-4 is bit harder,
since you have to sweet talk the federal government
into selling you some. Since RP-4 and JP-4 are both
basically different formulations of kerosene, it is
probably easier to just buy regular kerosene and deal
with a slight reduction in rocket engine performance.
When it comes to choosing a oxidizer/fuel
combination, the primary decision seems to
be whether to tackle cryogenic temperatures
or not:
-
Cryogenics
-
If the answer to the cryogenic
question is `yes', liquid oxygen (LOX) is
the obvious oxidizer choice. Once LOX has
been chosen, the next choice is whether to
deal with cryogenic temperature fuels. This
choice is not nearly as simple:
-
Cryogenic Fuel
-
Propane seems to be the best choice for
a cryogenic fuel -- it will simplify
tank design issues because it can be kept
at the same temperature as the LOX.
The drawback with propane is that it is
not as dense as hydrocarbon fuels (i.e.
gasoline, kerosene, methyl alcohol, etc.)
The lower density means that the fuel tank
will be larger and heavier; this could offset
the advantages of keeping the cryogenic
fuel and oxidizer at the same temperature.
-
Non-cryogenic Fuel
-
If a non-cryogenic fuel is chosen, gasoline
will probably edge out the other hydrocarbon
fuels based on cost and availability
considerations.
It is interesting to note that most commercial
launch vehicles seem to go for the LOX oxidizer
and either hydrocarbon or liquid hydrogen fuel.
The hydrocarbon fuel choice seems to be based
on density issues and the liquid hydrogen fuel
seems to be based on the excellent performance
that liquid hydrogen can provide. The LOX/Propane
combination seems to be largely unexplored.
-
No Cryogenics
-
If the decision concerning cryogenics is "no",
the oxidizer choice is going to be between
hydrogen peroxide and nitrous oxide. It is
looking like nitrous oxide is easier to
obtain than hydrogen peroxide, so that is
probably the oxidizer of choice. When it
comes to ambient temperature fuels, gasoline
still looks like it edges out the others
in terms of price and availability.
So what are the fuel/oxidizer combinations? The
following seem to be reasonable combinations that
are suitable for the amateur rocket community:
-
LOX/Propane
-
LOX/Propane seems to be a pretty good
choice for when both fuels and oxidizers
are cryogenic. This pair is going to
require figuring out how to cool down
the propane from ambient temperature
to cryogenic temperatures. This should
be no more difficult that running a
tube through a vat of liquid nitrogen.
-
LOX/Gasoline
-
Kerosene can be substituted for gasoline.
This pair only requires mastering the
handling of LOX. In addition, keeping
the LOX from freezing the fuel by
accident needs to be addressed.
-
Hydrogen Peroxide/Gasoline
-
This is one of two ambient temperature
fuel/oxidizer combinations. Again,
kerosene can be substituted for gasoline.
This pair requires mastering how to
generate hydrogen peroxide. Hydrogen
peroxide does have a higher performance
than nitrous oxide.
-
Nitrous Oxide/Gasoline
-
As usual kerosene can be substituted
for gasoline. The rocket will need
to be bigger and heavier to offset the
lower performance of nitrous oxide.
Both the fuel and the oxidizer need to be stored in
tanks. Each tank has to be able to withstand the
entire weight of all of the stages above it plus
the fluid in the tank (i.e. fuel or oxidizer)
while the rocket is accelerating at multiple G's
of acceleration. Furthermore, the LOX tanks will
need to have enough insulation to prevent undue
`boiling' in the LOX. Given the number of other
mass compromises that are going to be necessary
to build an amateur orbital rocket, tank mass
will have to pretty comparable with rockets made
by the aerospace community.
The lightest tanks are made out of composites,
which are also expensive to manufacture.
Aluminum is probably the only relatively affordable
tank material for the amateur rocket community.
Even so, an orbital rocket built out of aluminum
is a lot of aluminum and minimizing the amount
of aluminum required will significantly reduce
the entire cost of the rocket.
The only real `trick' I know of to reduce tank
mass is to keep it pressurized. This technique
is used by the Centaur upper stage. The idea
here can be demonstrated using an almost empty
plastic 2 liter bottle of carbonated soda. When
the cap is off, the bottle is extremely flexible.
When you put the cap on and shake the bottle, it
becomes pressurized and much more rigid. By
pressurizing a rocket tank, far less aluminum is
needed.
Pressurization is typically accomplished by storing
some helium in some high strength titanium spheres.
An amateur rocket may have to compromise by using
a somewhat larger helium storage tank at a
correspondingly lower pressure.
One disadvantage of pressurized tanks is that if
pressurization is ever lost while the tanks has
weight on it, the tank tends to crumple up. I
think the amateur rocket community can live with
this constraint.
{Talk about flat end-plates vs. spherical end-plates.}
{Talk about inter-stage adaptors.}
In general, the fluid with the highest density
is put in the lower tank to lower the vehicle
center of gravity. Since oxidizers are almost
always denser than the corresponding fuel, this
means that the oxidizer thank is usually on the
bottom.
A feed line needs to be run from the upper tank
to the lower tank. There are three basic
places to run the feed line:
-
Down the center
-
xxxx
-
On the outside
-
xxx
-
{Talk about the fuel line placement.}
Assembling a tank out of aluminum is no easy task.
The ideal solution would be to find a source of
extruded aluminum tubing that is the right length
and thickness and just weld two end caps on either
end. If no extruded aluminum is available, sheet
aluminum must be purchased, cut, shaped and welded
into a tube of the appropriate diameter. The more
welding there is, the more chances there are to
make a mistake that will compromise tank integrity.
{Talk about tank slosh.}
{Talk about tank assembly.}
{Talk about tank testing.}
{Talk about tank insulation.}
Engines
Developing a liquid rocket engine is no small task.
Developing a cheap liquid rocket engine makes the
task even harder still.
There is a wonderful book called entitled
How to Design, Build and Test Small Rocket
Engines
[Krzycki67] that is basically a `must read' for
any one contemplating the development of an inexpensive
liquid rocket engine. Through the hard work of Daniel
Risacher and some others, this book is now available on
the
web. The overall strategy espoused by the book is:
-
Be safe! Operate the engine remotely and
from behind a blast protection wall.
-
Start small with engines in the 10 to 25 pound
thrust range.
-
Start with gaseous oxygen before attempting
to work with liquid oxygen. Kerosene or
gasoline seemed to be the initial fuel choice.
-
Manufacture the initial engines out of copper
and use water cooling.
While their strategy makes sense as far as it goes,
it does not result in flight ready liquid rocket
engines.
Many years later, I read an abstract in NASA Technical
Briefs that suggested an interesting way of
constructing rocket engines. The abstract proposed
constructing an engine out of a stack of machined
metal sheets. Each sheet in the stack would be
individually machined to have holes for the
nozzle or combustion chamber along with holes for
regenerative cooling. Basically the whole assembly
would be bolted together. It occurs to me that this
might be the base technology for building cheap
liquid rocket engines.
{Talk about titanium plates versus aluminum plates.}
{Lots more goes here.}
Unlike hypergolic fuels, when you mix liquid oxygen and
kerosene, they do not spontaneously ignite. Thus, some
form of ignitor is needed. A fairly reasonable choice
for an ignitor is to use have port in the injector plate
through which the exhaust gases of an Estes model rocket
engine can shot. Reliable electric ignition of a model
rocket engine no longer seems to be a problem.
{Put a picture here.}
Liquid rocket engines need some sort of pump to take
the low pressure fuel and oxidizer, pressurize them, and
force them into the relatively high pressure combustion
chamber. In general, the higher the combustion chamber
pressure, the higher the overall rocket engine performance.
The aerospace companies tend to use turbo pumps for rocket
engines. While turbo pumps are extremely powerful for their
overall mass, they are difficult to design, manufacture,
and test. In one space propulsion class I audited at MIT,
we estimated a full 10% of the energy of a liquid rocket
engine is used to run the turbo pumps to pressurize the
fuel. An amateur rocket that uses a liquid rocket
engine is going to need to expend a similar amount of
energy for pressurization and be inexpensive as well.
A pump based on pistons is a serious possibility as
is shown in the diagram below:
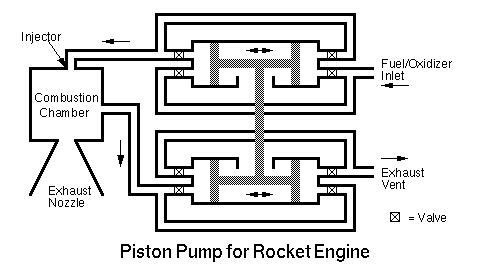
The basic idea is similar to how a piston is driven
back and forth on a steam locomotive. Hot gases from
the combustion chamber are alternately routed from one
side of the piston to the other via valves. Other
valves are alternately opened and closed to vent the
combustion cases when they are no longer needed. A
mechanical linkage is used to drive another piston back
and forth to pressurize the fluid (i.e. fuel or oxidizer.)
Again, valves are alternately opened and closed to route
the fluid into and out of the pressurization cylinders.
Since there is a pressure drop across the injector as
the fluid is injected into the combustion chamber, it
is necessary to construct the pump so that it delivers
the fluid at a higher pressure than the combustion
chamber. Since the force on the mechanical linkage
is the direct product of the pressure times the piston
area, it is possible to write the following balance
equation:
Pcc Acc = Pf Af
where
-
Pcc
-
is the combustion chamber pressure measured
in Pascals (i.e. Nt/M2),
-
Acc
-
is the area of the piston being driven by
the combustion chamber gases measured in
square meters,
-
Pf
-
is the fluid (i.e. fuel or oxidizer) delivery
pressure to the combustion chamber injector
measured in Pascals, and
-
Af
-
is the area of the fluid piston measured in
square meters.
Since Pf > Pcc, it follows that Af < Acc.
The development of a workable piston pump for amateur
liquid rocket motors will have to overcome many
difficulties, some of which are listed below:
-
Dealing with Hot Combustion Gases
-
Since the gases that are coming out of the
combustion chamber are quite hot, it can be
quite challenging to build a set of pistons
and valves that can operate at such high
temperatures. My suggestion is to cool
he combustion chamber gases down some with
a simple `heat exchanger' until they are at
a more workable temperature. All I am thinking
of when I say `heat exchanger' is a tube with
hot exhaust gases on one side and liquid
oxygen running past it on the other side.
Once the temperatures are in the same region
as automobile engine exhaust gases, they
should be workable by amateurs again.
-
High Pressure Differential Pistons
-
Since building a piston that can withstand
several the tens of atmospheres of pressure
differential can be quite challenging, it
may be desirable to build multi-stage pumps
to reduce the pressure differential across
each piston.
-
Induced Combustion Instability Due to Valve Operation
-
Each time a valve is opened or closed, there
will a corresponding increase or decrease in
the down stream pressure. These pressure
variations can cause combustion instabilities
in the combustion chamber. Obviously, if the
combustion instabilities are bad enough, they
can cause the engine to explode. A possible
solution to this problem is to build the pumps
so that they have parallel stages that operate
out of phase with one another.
-
{More issues here}
-
...
{More about pumps goes here.}
If there is any hope of getting into orbit, it is
going to be necessary to steer the rocket into
orbit. Rocket steering is accomplished by thrust
vector control.
The most efficient form of thrust vector control
is to put the entire rocket engine on gimbals
and move it to and fro. Building gimbals that
withstand the large forces needed to get a rocket
going are not easy. Furthermore, if the rocket
engine gimbals, it is necessary to have flexible
fuel and oxidizer lines. A flexible fuel hose
can be made out of rubber. A flexible oxidizer
line is a little more challenging because of the
cryogenic temperatures involved.
One possible solution to the thrust vector control
problem is to use use the exhaust gases from the
pumps. Using valves the pump exhaust gases can be
directed and vented to push the rocket to and fro.
It is unclear to me whether there are enough gases
available to generate enough side thrust to actually
steer the rocket.
Another possible solution is to uses clusters of
three or more off axis rocket engines for thrust
vector control. If the engines are 2-3 degrees
off axis, a small amount of throttling in the
engines can be used to create asymmetric thrust
to steer the rocket. This requires that the
rocket engines have some throttle capability.
{Talk about using GPS instead of inertial navigation.}
{Navigation section goes here.}
{Talk about using the Iridium constellation as
telemetry option.}
{Telemetry section goes here.}
{Launch facilities section goes here.}
{Summary goes here.}
-
[Crisalli97]
-
700 Miles North... ...and 50 Miles Up?
by David E. Crisalli in
High Powered Rocketry,
Vol. 12 No. 8, (Oct./Nov Issue) 1997
published by
Tripoli Rocketry Association,
P.O. Box 280 Bessemer, AL USA 35021
Phone: 1-205-424-8357
ISSN: 1070-5244.
-
[Krzycki67]
-
How to Design, Build, and Test Small
Liquid-Fuel Rocket Engines by
Leroy J. Krzycki, 1967.
ISBN: 9600-1980-4. URL:
http://www2.jps.net/~gramlich/projects/rocket/index.html.
-
[Lide96]
-
CRC Handbook of Chemistry and Physics,
77th Edition 1996-1997
David R. Lide, Editor-in-Chief, 1996
published by
CRC Press, Inc.,
9000 Corporate Blvd.,
Boca Raton, FL 33431.
ISBN: 0-8493-0477-6
Library of Congress Card Number: 13-11056
Copyright (c) 1995, 1998, 2001
Wayne Gramlich.
All rights reserved.
